Mutant NPM1 as target for immunotherapy of acute myeloid leukemia
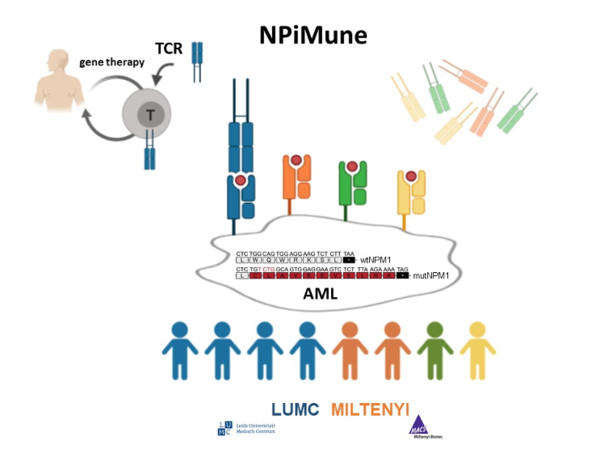
Acute myeloid leukemia (AML) is the most common type of acute leukemia in adults. Approximately 18,000 people in Europe and 700 patients in the Netherlands are diagnosed with AML each year. AML is treated by chemotherapy, which can be followed by allogeneic stem cell transplantation. Although most patients initially respond, relapses often occur. Nucleophosmin (NPM1) is a gene that is mutated in one third of AML. LUMC and MILTENYI collaborate on development of T cell receptor (TCR) gene therapy to target mutant NPM1 on AML.
At the LUMC, a TCR for mutant NPM1 has been found that can be used to treat 15% of AML patients. Together with MILTENYI, this TCR is currently under development to test its toxicity and efficacy in a clinical study in 2020/2021. Core element is MILTENYI’s CliniMACS Prodigy platform which allows automated and de-centralised manufacturing in a closed system, thereby reducing economic burden on healthcare systems in addition to offering potential cure to the individual patient.
In the collaborative research, the TCR for mutant NPM1 that is known to be effective against AML will be tested in the lentivirus as used in the clinical trial and analysed for potential toxicity. In addition, another TCR for a different NPM1 structure will be analyzed for efficacy. If effective, this TCR will also be tested in the lentivirus and analysed for toxicity. Finally, new T cells for mutant NPM1 will be isolated and their TCRs will be similarly analysed.
In conclusion, LUMC and MILTENYI collaborate on clinical development of a TCR for mutant NPM1 that can be used to treat 15% of AML patients. In addition, other TCRs for mutant NPM1 will be isolated and tested with the aim to increase the population that can be treated with gene therapy to up to one third of AML patients.