Detection of protein-aggregates for differential diagnosis of dementias by RT-DEM
Researchers from VUmc along with industrial partners – ADx Neuroscience aim to develop a novel assay called RT-DEM, which will be a first method in the field of dementia to be able to detect and distinguish suspected patients based on their specific tau pathologies. To establish the assays, they will first detect different Tau isoform -aggregates in post-mortem brain tissue and post-mortem CSF samples of all tauopathies. To clinically validate the use of this approach for in-vivo diagnosis, the test will be optimised to detect seeds in clinical CSF obtained by lumbar puncture of living-patients. A dedicated effort will also be made to differentiate between different human Tauopathies (AD, AGD, FTD, CBD and PSP) by further optimising the overall sensitivity and specificity of the method.
Implementation of such a test in the Netherlands will be of great help for early diagnosis of dementia subtypes associated with tauopathies. The cost of all brain diseases combined amount to one third of total health care costs (source: Hersenstichting). The cost of dementia amounted to € 4 billion in 2013, 5% of total health care costs in the Netherlands. An early diagnosis could therefore reduce the healthcare costs and this has a favorable/positive economic impact. Once the test has been established and validated for ante-mortem diagnosis of dementia patients, they can start with the implementation in clinical practice. For this project, they can count on the involvement of the Neurologists at the VU Medical Center. They can apply the test on clinical patients in order to verify the method’s scope and expand the clinical validation. This will not only give them confidence to perform the test in clinical routine but they can further disseminate the knowledge in the relevant medical field.
Development of RT-DEM test for detection of Tauopathies got delayed due to hurdles in obtaining and generating a good quality tau substrate. However, the tau construct was arranged from the collaborators in the USA and have set up the lab facilities to perform recombinant tau expression and purification in-house in order to develop a tau detection test with high reproducibility. In the meanwhile, the efforts were focused to apply the principle of RT-DEM for detection of Synucleinopathies including Parkinson’s and Lewy Body Dementia and even some highly reproducible and promising results have been obtained in tissue/CSF, patient types. In the future, the aim is to develop RT-DEM for both Synucleinopathies as well as Tauopathies.
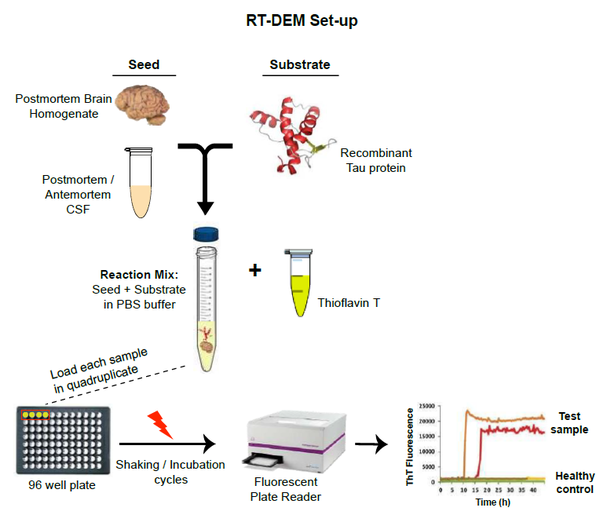
Figure 1: In RT-DEM, the pathogenic seeds present in a test sample induce amyloid fibril forma -tion of bacterially expressed recombinant protein which is used as a substrate. The resulting amy-loid fibrils are then detected by the enhanced fluorescence of the amyloid-sensitive dye, thioflavin T (ThT) in a fluorescent plate reader. Repeated cycles of shaking / incubation are used to exponen-tially increase initial low levels of pathogenic seed, facilitating a greater sensitivity of detection. Thus, If the test sample contains an infectious seed, it will cause the recombinant protein to aggre-gate and make thioflavin-T fluoresce.


